|
MARIUS DAGYS Senior Researcher |
 |
Bioelectrochemical Systems in Biosensors and Bioreactors
Mediated and direct electron transfer (ET) coupling of enzymes to electrodes is important in realizing bioelectrocatalysis, which is often exploited as a basic principal of biosensors, biofuel cells, and other bio-based devices. These technologies exploit the inherent enzyme substrate specificity, for example, enzyme-based biosensors excel in direct measurement of single compound in presence of interfering materials in complex media such as blood. On the other hand, if the power density generated by enzyme-based electrode is high enough, biofuel cells can be constructed, where bioelectrodes selectively oxidise and reduce abundant fuel (i.e. glucose and oxygen) and provide electric power for implantable devices. The fragile nature of proteins dictates that the electrochemical properties of such biodevices degrade over time. Therefore, a number of techniques are developed to protect the biomolecule and extend the working period of device. The shortcoming could be avoided whatsoever by adsorbing live, whole cells on electrodes at the expense of reduced power density.
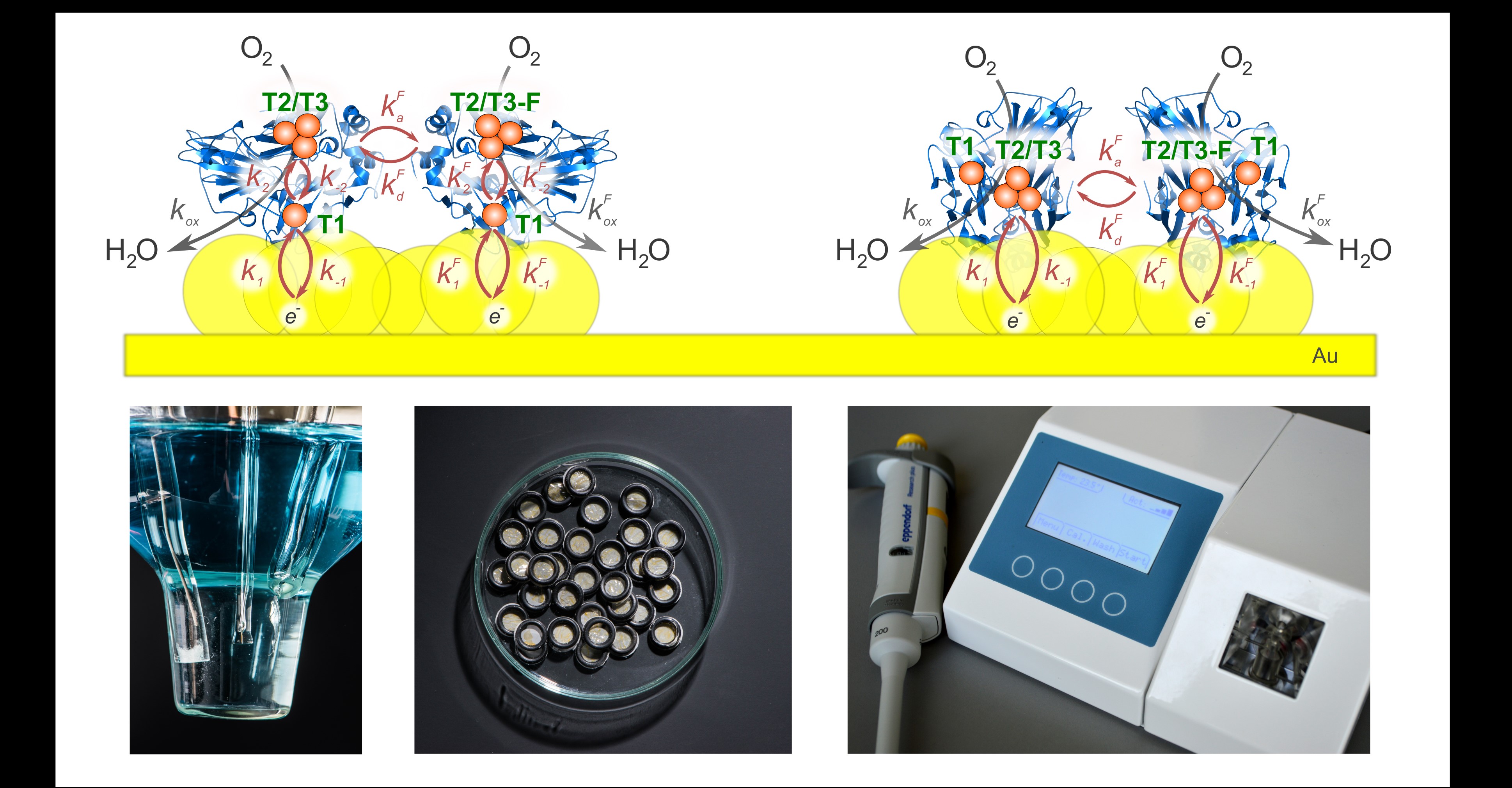
Our team is proficient at constructing bioelectrochemical systems by wiring oxidoreductases to gold and carbon based electrode surfaces [1–3]. Our team is also developing bioreactor systems, where wasteful saccharide substrates are selectively oxidised and high-value oxidation products are produced. For such an approach, we utilize bi-enzymatic reaction with biosensor-based microprocessor-controlled substrate dispensing. In order to obtain a self-regulating system, the fluid dispensing and sensor devices are coordinated by an advanced algorithm embedded in microcontroller-based electronic system. All the custom components were designed and produced by our team. Recently, molecularly imprinted polymers based on polypyrrole and polyaniline preparations have intensively been studied for sensor electrode application [4]; the approach should help in finding new ways to discover new supramolecular systems for small biomolecule detection.
SELECTED PUBLICATIONS
1. Ratautas, D., Dagys, M. Nanocatalysts containing direct electron transfer-capable oxidoreductases: recent advances and applications. Catalysts. 2020, 10: 9.
2. Gineitytė, J., Meškys, R., Dagys, M., Ratautas, D. Highly efficient direct electron transfer bioanode containing glucose dehydrogenase operating in human blood. J. Power Sources. 2019, 441: 227163.
3. Dagys, M., Laurynėnas, A., Ratautas, D. et al. Oxygen electroreduction catalysed by laccase wired to gold nanoparticle via the trinuclear copper cluster. Energy & Environmental Science. 2017, 10: 498.
4. Bagdžiūnas, G. Theoretical design of molecularly imprinted polymers based on polyaniline and polypyrrole for detection of tryptophan. Mol. Syst. Des. Eng. 2020, 5: 1504.
Dehydrogenases in Custom Sensor-Controlled Bioreactors
The use of the dehydrogenases to oxidize substrates in bioreactors is very attractive due to their broad substrate specificity and high catalytic activity. However, the application requires an effective mediated enzyme reoxidation method. The mediator in regeneration scheme must be highly reactive with the enzyme to regenerate; all forms of the mediator must be stable, nontoxic and cheap. The oxidized mediator form is produced in reaction with heme peroxidase, which exhibits high catalytic activity at pH 7.0 and broad substrate specificity. Peroxidase uses hydrogen peroxide as co-substrate to oxidase mediators. To avoid the hydrogen peroxide-induced enzyme inactivation, the addition of hydrogen peroxide to the reactor mixture was performed in very small doses of 40 nL by using the syringe pump developed by our team. The rate of dosage was controlled by analysing the data of our custom highly-sensitive hydrogen peroxide and optical, oxidized mediator-form sensors, all combined into microcontroller-coordinated control algorithm. As of today, the turnover number of PQQ glucose dehydrogenase in model reactor reaches ~1x107, which means one can produce a valuable product at ~0,65 Eur/g and expect to sell it for about a hundred times more (RCL grant No. 01.2.2-LMT-K-718-01-0019).

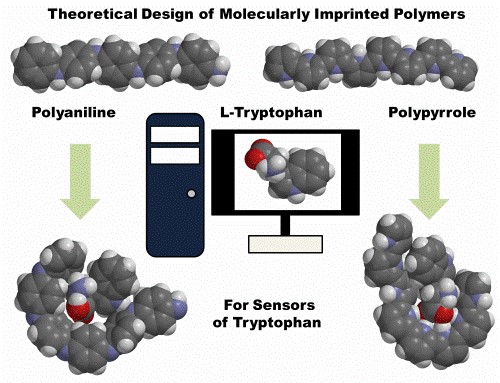
Theoretical Design of Molecularly Imprinted Polymers for Analysis
Creation of molecularly imprinted polymers (MIPs) as the supramolecular systems with tailor-made binding sites complementary to template molecules in shape, size and functional groups is an important task for the analytical, physical and theoretical chemistries. In this work, the polypyrrole and polyaniline-based host - guest MIPs were theoretically studied for the detection of tryptophan. These simulations showed that polyaniline is not suitable for the selective detection of tryptophan due to high flexibility of its chains and low energy of intermolecular interactions. In contrast, the polypyrrole-based hosts can be used to detect tryptophan, because all these simulated forms shaped the bow-shaped inner cavity with the strongly coordinated target molecule. Moreover, the insights will help in finding new ways to discover new supramolecular systems for small biomolecule detection [4] (RCL grant No. S-MIP-20-45).
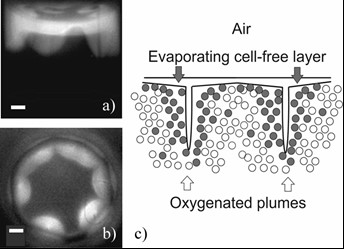
Self-Organization of Bacteria
Bioanalytical systems can be constructed by using whole-cell biosensors, where bacteria are grown on electrode surfaces. We use bioluminescence imaging to record images of liquid mixed cultures of the lux-gene reporter E. coli and other bacteria in microtiter plate wells and in vertical Hele-Shaw cells. Analysis of the experimental data together with mathematical modelling suggests the following interpretation of pattern formation (right figure: a) and b) show typical side-view and top-view images of cylindrical samples (bar – 1 mm), the scheme of a system that forms spatiotemporal patterns is shown in c). The evaporation- and settling-driven instability of the surface layer results in formation of oxygenated plumes. In the vicinity of the plumes, active cells (grey circles) ‘aggregate’ and ‘grow’ at the expense of passive cells (white circles). These studies were partly funded by RCL grant No. S-MIP-17-98.