About
Main research directions. Synthesis of prooxidant xenobiotics (quinones, aromatic nitrocompounds, N-oxides, etc.), studies of interaction of these compounds with flavoenzymes and their impact on their cytotoxicity, mechanisms of flavoenzyme catalysis, prooxidant cytotoxicity of polyphenolic compounds. Since 1992, the Department collaborates with research centers in Lithuania, Belarus, Great Britain, Spain, Italy, USA, New Zealand, the Netherlands, France, Sweden, and Ukraine. We have participated in projects supported by the EC (2), NATO (2), the Royal Swedish Academy of Sciences (1), bilateral projects with France (2), Belarus (1), and Ukraine (1), COST actions (4), received a number of grants from LSSSF and RCL including the project funded by European Social Fund (ESF) under the Human Resources Development Action Programme, the Global Grant measure (2011-2015).
Synthesis of prooxidant compounds. We have synthesized or resynthesized over 300 compounds of different groups – nitroaromatic and nitroheterocyclic compounds, aliphatic nitrates, aromatic N-oxides, quinones, benzofuroxans, etc. (Fig. 1), examined their electrochemical properties and characterized their reduction energetics by quantum-mechanical methods.
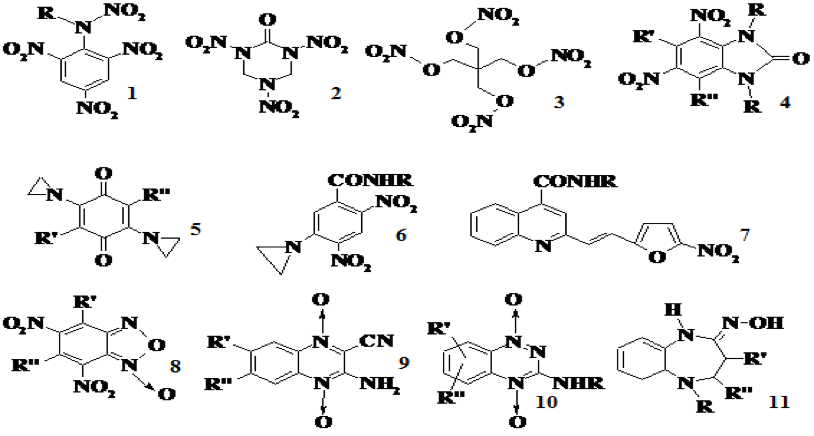
Fig. 1. High-energy substances tetryl (1), keto-RDX (2), PETN (3), and other groups of compounds: nitrobenzimidazolonesi (4), aziridinyl-substituted quinones (5) and nitrobenzenes (6), nitrofurans (7), benzofuroxanes (8), di-N-oxides of quinoxaline (9) and 1,2,4-benzotriazine (10), and heterocyclic oximes (11).
The mechanisms of single- and two-electron reduction of quinones, nitroaromatic compounds, and other prooxidants by flavoenzymes. Flavoenzymes electrontransferases reduce quinones, aromatic nitrocompounds and N-oxides into their free radicals. The latter are reoxidized by oxygen with the formation of superoxide and other reactive oxygen species (ROS) causing the oxidative stress which significantly determines their cytotoxicity , antitumour and other therapeutic activity of the compounds. The enzymatic single-electron reduction reactions of the compounds have been examined applying mammalian cytochrome P-450 reductase, NO-synthase (in collaboration with J.-L. Boucher, Universite de Paris V), Anabaena sp. and P. falciparum ferredoxin:NADP+ reductase (in collaboration with C. Gomez-Moreno, Universidad de Zaragoza, and A. Aliverti, Universita de Milano), and mixed single- and two-electron reduction reactions by using mammalian mitochondrial complex I (in collaboration with A.D. Vinogradov, Moscow University), apoptosis inducting factor (in collaboration with I.F. Sevrioukova, California University, Irvine), lipoamide dehydrogenase, plant and bacterial thioredoxin reductases (in collaboration with J.-P. Jacquot and N. Rouhier, Universite de Nancy), and bacterial flavohemoglobin (in collaboration with L. Baciou and F. Lederer, Universite de Paris Sud). The single-electron reduction of quinones and nitroaromatic compounds by aforementioned enzymes is described by Marcus' model, and the rates are weakly influenced by the structure and VdWvol of the compounds. In several cases, the redox states of the enzymes responsible for the rate-limiting stage have been identified. Mixed single- and two-electron reduction is characterized by e-,H+,e- mechanism determined by the destabilization of neutral flavin semiquinone.
Two-electron reduction by mammalian DT-diaphorase or bacterial nitroreductases decreases the cytotoxicity of simple quinone compounds, but increases the cytotoxicity of aziridinyl-substituted quinones and nitroaromatic compounds due to formation of DNA-alkylating products (Fig. 2).
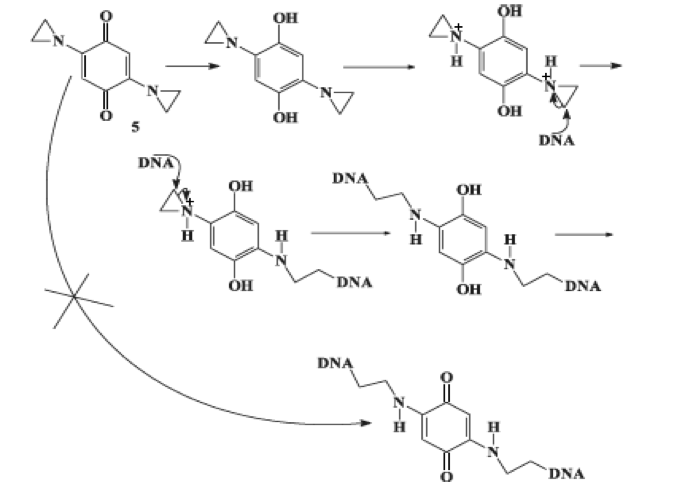
Fig. 2. Two-electron reduction of aziridinyl-substituted quinones with subsequent alkylation of DNA.
It has been examined the reduction of quinones and nitroaromatic compounds mediated by DT-diaphorase, E. cloacae nitroreductase B (in collaboration with R. Koder, Kentucky University), PETN reductase (in collaboration with N.S. Scrutton, Manchester University), and E. coli nitroreductase A (in collaboration with D.F. Ackerley, Welington University). These reactions have been found to be characterized by a strong substrate structure specificity because of significant conformational changes of the enzyme active sites and by negative ∆S≠. Depending on the stability of anionic flavin semiquinone radical, these reactions proceed through single- (H-) or three-step (e-,H+,e-) hydride transfer.
Mammalian cell culture cytotoxicity of quinones and nitroaromatic compounds. Applying several transformed and primary mammalian cell cultures (Center of Innovative Medicine), the cytotoxic action of quinones and nitroaromatics has been found to be mainly determined by the oxidative stress, and that the cytotoxicity of compounds increases with an increase in their single-electron reduction potential, with ∆log cL50/∆E17 of -8 – -10 V-1. Significantly increased cytotoxicity of aziridinyl-substituted quinones is attributed to their activation by DT-diaphorase. Aziridinyl-benzoquinone resistant cell sub-lines possess 10-fold decreased activity with DT-diaphorase and glutathione-S-transferase, whereas the activity of prooxidant and antioxidant enzymes varies in the limits of ±50%.
Quinones and nitroaromatic compounds as inhibitors and subversive substrates of antioxidant flavoenzymes. Quinones and nitroaromatic compounds inhibit the antioxidant mammalian and parasite flavoenzymes glutathione reductase (GR) and trypanothione reductase (TR), and mammalian thioredoxin reductase (TrxR), thus inhibiting the regeneration of –SH groups in the cell. In parallel, they may be reduced by the aforementioned enzymes in mixed single- and two-electron way, thus subverting their antioxidant functions. The above mechanisms may be employed in the design of new antiparasitic and anticancer agents. The interaction of the above compounds has been examined with erythrocyte and P. falciparum GR (in collaboration with E. Davioud-Charvet, Universite de Strasbourg), T. congolense TR (in collaboration with J. Blanchard, Albert Einstein College of Medicine, NY), and mammalian TrxR (in collaboration with E. Arner, Karolinska Institutet). A number of efficient inhibitors of the above enzymes have been discovered. It has been established that apart from the reduced flavin, the above compounds may be reduced by the catalytic SeH-SH-moiety of TrxR. In collaboration with P. Grellier (MNHN, Paris), the relation between the efficiency of inhibition of P. falciparum GR by quinones and nitroaromatic compounds and their in vitro antiplasmodial activity has been established.
Prooxidant cytotoxicity of polyphenols. The antioxidant activity of polyphenols (flavonoids, polihydroxybenzenes, etc.) relies on neutralization of ROS. Therefore they are considered as useful food components; their antitumour and antiparasitic activity have been examined. However, polyphenols possess prooxidant cytotoxicity, because during their (auto)oxidation, H2O2 and quinone/quinomethide oxidation products are formed which alkylate –SH groups in the cell. The cytotoxicity of polyphenols in several cell lines increases with a decrease in their single-electron oxidation potential (∆log cL50/∆E27 = 1 – 2 V-1 ), and with an increase in their lipophilicity. Hydroxylation and oxidative demethylation of polyphenols by cytochromes P-450 increases their cytotoxicity, whereas the methylation by catechol-O-methyltransferase decreases it. During the collaboration with P. Venskutonis (Kaunas Technological University), prooxidant cytotoxicity of a number of plant and herb polyphenolic extracts has been characterized.
Projects
Key grants 2012-2023
National Research Projects funded by Research Council of Lithuania
- Redox chemistry, biochemistry and cytotoxicity of aromatic nitrocompounds and N-oxides: new insighths (No. DOTSUT-34/09.33-LMT-K712-01-0058). Habil. dr. N. Čėnas. 2018-2022. Global grant measure, European Social Fund (ESF).
- Molecular mechanisms of toxicity and antitumor activity of quinones and polyphenols: enzymatic redox reactions, cytotoxicity, signal transduction and proteomics. Habil. Dr. N. Čėnas. 2011–2015. European Social Fund (ESF) under the Human Resources Development Action Programme, the Global Grant measure, project No. VP1-3.1-ŠMM-07-K-01-103.
- New generation N-heterocyclic quinones: rational synthesis and elucidation of anticancer activity (MIP-032/2014). Dr. Ž. Anusevičius. 2014-2016.
- Heterocyclic N-oxides: synthesis, cytotoxicity, and interaction with target enzymes (MIP-080/2011). Dr. J. Šarlauskas. 2011-2012.
International Research Projects
- COST Action CM0801. New drugs for neglected diseases. Dr. J. Šarlauskas. Habil. Dr. N. Čėnas. 2009-2012.
- COST Action CM1307. Targeted chemotherapy towards diseases by endoparasites. Dr. J. Šarlauskas. Habil. Dr. N. Čėnas. 2014-2017.
- Bilateral Lithuanian-French programme Gilibert. Quinones and nitroaromatic compounds as the subversive substrates of flavohemoglobins: mechanisms and biomedicinal implications (No. TAP LZ 07/2013). Habil. Dr. N. Čėnas 2013-2014.
- Bilateral Lithuania-Ukraine programme. Investigation of L- and D-lactate: cytochrome с oxidoreductases isolated from the recombinant yeast Hansenula polymorpha and their usage for construction of amperometric biosensors (No. TAP LU 03/2014). Dr. K. Krikštopaitis. 2014-2015.
- Bilateral Lithuania-Belarus programme. The characterization of the inter-protein interaction and the modulators of the redox equivalent transfer in steroid hydroxylation systems (No. TAP LB 02/2013). Dr. Ž. Anusevičius. 2013-2014.
Main publications
2022
- Lesanavičius M., J.L. Boucher, N. Čėnas. 2022. Reactions of recombinant neuronal nitric oxide synthase with redox cycling xenobiotics: a mechanistic study. International Journal of Molecular Sciences 23(2): 980.
- Lesanavičius M., D. Seo, N. Čėnas. 2022. Thioredoxin reductase-type ferredoxin: NADP(+) oxidoreductase of Rhodopseudomonas palustris: potentiometric characteristics and reactions with nonphysiological oxidants. Antioxidants 11(5): 1000.
- Misevičienė L., K. Krikštopaitis, N. Čėnas. 2022. The comparative study of redox properties of recombinant human cytosolic and mitochondrial NADPH:thioredoxin reductases. Chemija 33 (2): 40-45.
- Polmickaitė-Smirnova E., I. Buchovec, S. Bagdonas, E. Sužiedelienė, A. Ramanavičius, Ž. Anusevičius. 2022. Photoinactivation of Salmonella enterica exposed to 5-aminolevulinic acid: Impact of sensitization conditions and irradiation time. Journal of Photochemistry and Photobiology B-Biology 231: 112446.
- Šarlauskas J., J. Stankevičiūtė, J. Tamulienė. 2022. An efficient synthesis and preliminary investigation of novel 1,3-dihydro-2h-benzimidazol-2-one nitro and nitramino derivatives Materials 15(23): 8330.
- Šarlauskas J., J. Tamulienė. 2022. Preparation and characterization of cationic energetic salts of 5-amino-3-[(2,4,6-trinitrophenyl)amino]-1H-1,2,4-triazole (APATO). Central European Journal of Energetic Materials 19(3): 311-325.
- Šarlauskas J., K. Tulaitė, J. Tamulienė. 2022. Investigation of oxygen influence to the optical properties of tirapazamine. Journal of Molecular Modeling 28(4): 96
- Uebe J., Z. Kryževičius, A. Kuhan, A. Torkelis, L. Kosychova, A. Žukauskaitė. 2022. Improving of pyrolysis oil from macroalgae Cladophora glomerata with HDPE pyrolysis oil. Journal of Marine Science and Engineering 10(2): 131. IF 2,744
- Uebe J., Z. Kryževičius, R. Majauskienė, M. Dulevičius, L. Kosychova, A. Žukauskaitė. 2022. Use of polypropylene pyrolysis oil in alternative fuel production. Waste Management & Research 0734242X211068243.
2021
- Čėnas N., A. Nemeikaitė-Čėnienė, L. Kosychova. 2021. Single- and two-electron reduction of nitroaromatic compounds by flavoenzymes: mechanisms and implications for cytotoxicity. International Journal of Molecular Sciences 22 (16): 8534
- Cichocki B. A, M. Donzel, K. C. Heimsch, M.Lesanavičius, L. Feng, E.J. Montagut, K. Becker, A.Aliverti, M. Elhabiri, N. Čėnas, E. Davioud-Charvet. 2021. Plasmodium falciparum ferredoxin-NADP+ reductase-catalyzed redox cycling of plasmodione generates both predicted key drug metabolites: implication for antimalarial drug development. Infectious Diseases 7(7) 1996-2012.
- Nemeikaitė-Čėnienė A., A. Marozienė, L. Misevičienė, J. Tamulienė, A. Yantsevich, N. Čėnas. 2021. 5Flavoenzyme-catalyzed single-electron reduction of nitroaromatic antiandrogens: implications for their cytotoxicity. Free Radical Research 55(3): 246-254
- Šarlauskas J., J. Tamulienė, S. Bekešienė, A. Kravcov. 2021. Benzimidazole derivatives as energetic materials: A theoretical study. Materials 14 (15): 4112.
2020
- Benitez D., M. A. Comini, Ž. Anusevičius, J. Šarlauskas, V. Miliukienė, E. Miliuvienė, N. Čėnas. 2020. 5-Vinylquinoline-substituted nitrofurans as inhibitors of trypanothione reductase and antitrypanosomal agents. Chemija 31: 111-117.
- Lesanavičius M., A. Aliverti, J. Šarlauskas, N. Čėnas. 2020. Reactions of Plasmodium falciparum Ferredoxin:NADP+ Oxidoreductase with Redox Cycling Xenobiotics: A Mechanistic Study. International Journal of Molecular Sciences 21:3234
- Nemeikaitė-Čėnienė, J. Šarlauskas, V. Jonušienė, L. Misevičienė, A. Marozienė, A. Yantsevich, N. Čėnas. 2020. QSARs in prooxidant mammalian cell cytotoxicity of nitroaromatic compounds: the roles of compound lipophilicity and cytochrome P-450- and DT-diaphorase-catalyzed reactions. Chemija 31: 170-177.
- Nemeikaitė-Čėnienė, J. Šarlauskas, L. Misevičienė, A. Marozienė, V. Jonušienė, M. Lesanavičius, N. Čėnas. 2020. Aerobic cytotoxicity of aromatic N-oxides: the role of NAD(P)H:quinone oxidoreductase (NQO1). International Journal of Molecular Sciences 21:8754.
- Polmickaitė-Smirnova E., J. Šarlauskas, K. Krikštopaitis, Ž. Lukšienė, Z. Staniulytė, Ž. Anusevičius. 2020. Preliminary investigation of the antibacterial activity of antitumor drug 3-amino-1,2,4-benzotriazine-1,4-dioxide (tirapazamine) and its derivatives. Applied Sciences 10: 4062.
- Valiauga B., N. Rouhier, J.-P. Jacquot, N. Čėnas. 2020. Characterization of redox properties of FAD cofactor of Thermotoga maritima thioredoxin reductase. Chemija 31: 191-195.
- Petkevičius V., J. Vaitekūnas, D. Tauraitė, J. Stankevičiūtė, D. Vaitkus, J. Šarlauskas, N. Čėnas, R. Meškys. Whole-Cell Biocatalysis Using PmlABCDEF Monooxygenase and Its Mutants: A Versatile Toolkit for Selective Synthesis of Aromatic N-Oxides. In Applied Biocatalysis: The Chemist’s Enzyme Toolbox, 528-533. Ed. J. Whittall, P. Sutton, John Wiley&Sons Ltd.
2019
- Marozienė A., M. Lesanavičius, E. Davioud-Charvet, A. Aliverti, P. Grellier, J. Šarlauskas, N. Čėnas. 2019. Antiplasmodial activity of nitroaromatic compounds: correlation with their reduction potential and inhibitory action on Plasmodium falciparum glutathione reductase. Molecules 24: 4509. Nemeikaitė-Čėnienė, A., Šarlauskas, J., Jonušienė, V., Marozienė, A., Misevičienė, L., Yantsevich, A.V. and Čėnas, N., 2019. Kinetics of Flavoenzyme-Catalyzed Reduction of Tirapazamine Derivatives: Implications for Their Prooxidant Cytotoxicity. International journal of molecular sciences, 20(18), p.4602.
-
Petkevičius, V., Vaitekūnas, J., Tauraitė, D., Stankevičiūtė, J., Šarlauskas, J., Čėnas, N. and Meškys, R., 2019. A Biocatalytic Synthesis of Heteroaromatic N‐Oxides by Whole Cells of Escherichia coli Expressing the Multicomponent, Soluble Di‐Iron Monooxygenase (SDIMO) PmlABCDEF. Advanced Synthesis & Catalysis.
- Petkevičius, V., Vaitekūnas, J., Vaitkus, D., Čėnas, N. and Meškys, R., 2019. Tailoring a Soluble Diiron Monooxygenase for Synthesis of Aromatic N-oxides. Catalysts, 9(4), p.356.
-
Polmickaitė-Smirnova, E., Bagdonas, S. and Anusevičius, Ž., 2019. Sensitization of Salmonella enterica with 5-aminolevulinic acid-induced endogenous porphyrins: a spectroscopic study. Photochemical & Photobiological Sciences.
-
Rekovič L., L. Kosychova, I. Bratkovskaja, R. Vidžiūnaitė. 2019. Synthesis and spectral characterization of novel 1,5-benzodiazepine oxime derivatives. Journal of the Serbian Chemical Society 84: 343-353.
-
Šarlauskas J., E. Polmickaitė-Smirnova, N. Čėnas, K. Krikštopaitis, Ž. Anusevičius. 2019. The QSAR study for antibacterial activity of structurally diverse nitroaromatics. Chemija 30: 41-48.
2018
- Valiauga, B., Misevičienė, L., Rich, M., Ackerley, D., Šarlauskas, J. and Čėnas, N., 2018. Mechanism of Two-/Four-Electron Reduction of Nitroaromatics by Oxygen-Insensitive Nitroreductases: The Role of a Non-Enzymatic Reduction Step. Molecules, 23(7), p.1672.
- Moussaoui, M., Misevičienė, L., Anusevičius, Ž., Marozienė, A., Lederer, F., Baciou, L. and Čėnas, N., 2018. Quinones and nitroaromatic compounds as subversive substrates of Staphylococcus aureus flavohemoglobin. Free Radical Biology and Medicine, 123, pp.107-115.
2017
- Valiauga, B., Williams, E.M., Ackerley, D.F. and Čėnas, N., 2017. Reduction of quinones and nitroaromatic compounds by Escherichia coli nitroreductase A (NfsA): characterization of kinetics and substrate specificity. Archives of biochemistry and biophysics, 614, pp.14-22.
- Smutok, O., Karkovska, M., Serkiz, R., Vus, В., Čenas, N. and Gonchar, M., 2017. A novel mediatorless biosensor based on flavocytochrome b 2 immobilized onto gold nanoclusters for non-invasive L-lactate analysis of human liquids. Sensors and Actuators B: Chemical, 250, pp.469-475.
- Peciukaityte-Alksne, M., Šarlauskas, J., Miseviciene, L., Maroziene, A., Cenas, N., Krikštopaitis, K., Staniulyte, Z. and Anusevicius, Ž., 2017. Flavoenzyme-mediated reduction reactions and antitumor activity of nitrogen-containing tetracyclic ortho-quinone compounds and their nitrated derivatives. EXCLI journal, 16, p.663.
- Šarlauskas, J., Tamulienė, J. and Čėnas, N., 2017. Aziridinyl-substituted benzo-1, 4-quinones: A preliminary investigation on the theoretical and experimental studies of their structure and spectroscopic properties. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 178, pp.136-141.
- Jarasiene-Burinskaja, R., Alksne, M., Bartuskiene, V., Voisniene, V., Burinskij, J., Cenas, N. and Bukelskiene, V., 2017. Study of the cytotoxic effects of 2, 5-diaziridinyl-3, 6-dimethyl-1, 4-benzoquinone (MeDZQ) in mouse hepatoma cells. EXCLI journal, 16, p.151.
2016
- Šarlauskas, J., Pečiukaitytė-Alksnė, M., Misevičienė, L., Marozienė, A., Polmickaitė, E., Staniulytė, Z., Čėnas, N. and Anusevičius, Ž., 2016. Naphtho [1′, 2′: 4, 5] imidazo [1, 2-a] pyridine-5, 6-diones: Synthesis, enzymatic reduction and cytotoxic activity. Bioorganic & medicinal chemistry letters, 26(2), pp.512-517.
- Ger, M., Kaupinis, A., Nemeikaite-Ceniene, A., Sarlauskas, J., Cicenas, J., Cenas, N. and Valius, M., 2016. Quantitative proteomic analysis of anticancer drug RH1 resistance in liver carcinoma. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics, 1864(2), pp.219-232.
- Stulpinas, A., Imbrasaitė, A., Krestnikova, N., Šarlauskas, J., Čėnas, N. and Kalvelytė, A.V., 2015. Study of bioreductive anticancer agent RH-1-induced signals leading the wild-type p53-bearing lung cancer A549 cells to apoptosis. Chemical research in toxicology, 29(1), pp.26-39.
2015
- Valiauga, B., Rouhier, N., Jacquot, J.P. and Čėnas, N., 2015. Quinone-and nitroreductase reactions of Thermotoga maritima thioredoxin reductase. Acta Biochimica Polonica, 62(2), pp.303-309.
- Kosychova, L., Karalius, A., Staniulytė, Z., Sirutkaitis, R.A., Palaima, A., Laurynėnas, A. and Anusevičius, Ž., 2015. New 1-(3-Nitrophenyl)-5, 6-dihydro-4H-[1, 2, 4] triazolo [4, 3-a][1, 5] benzodiazepines: Synthesis and Computational Study. Molecules, 20(4), pp.5392-5408.
2014
- Miliukienė, V., Nivinskas, H. and Čėnas, N., 2014. Cytotoxicity of anticancer aziridinyl-substituted benzoquinones in primary mice splenocytes. Acta Biochimica Polonica, 61(4), pp.833-836.
- Šarlauskas, J., Misevičienė, L., Marozienė, A., Karvelis, L., Stankevičiūtė, J., Krikštopaitis, K., Čėnas, N., Yantsevich, A., Laurynėnas, A. and Anusevičius, Ž., 2014. The study of NADPH-dependent flavoenzyme-catalyzed reduction of benzo [1, 2-c] 1, 2, 5-oxadiazole N-oxides (Benzofuroxans). International journal of molecular sciences, 15(12), pp.23307-23331.
2013
- Anusevičius, Ž., Nivinskas, H., Šarlauskas, J., Sari, M.A., Boucher, J.L. and Čėnas, N., 2013. Single-electron reduction of quinone and nitroaromatic xenobiotics by recombinant rat neuronal nitric oxide synthase. Acta Biochimica Polonica, 60(2).
- Šarlauskas J., A. Nemeikaitė-Čėnienė, L. Misevičienė, K. Krikštopaitis, Ž. Anusevičius, N. Čėnas. 2013. Redox properties and prooxidant cytotoxicity of a neuroleptic agent 6,7-dinitrodihydroquinoxaline-2,3-dione (DNQX). Acta Biochimica Polonica 60: 227-231.
Personnel
Head of department
Habil. dr. Narimantas Čėnas
Chief research fellow
phone: +370 5 223 4392
email:
Research staff
PhD students
Technical staff
| Svetlana Belik technician |
